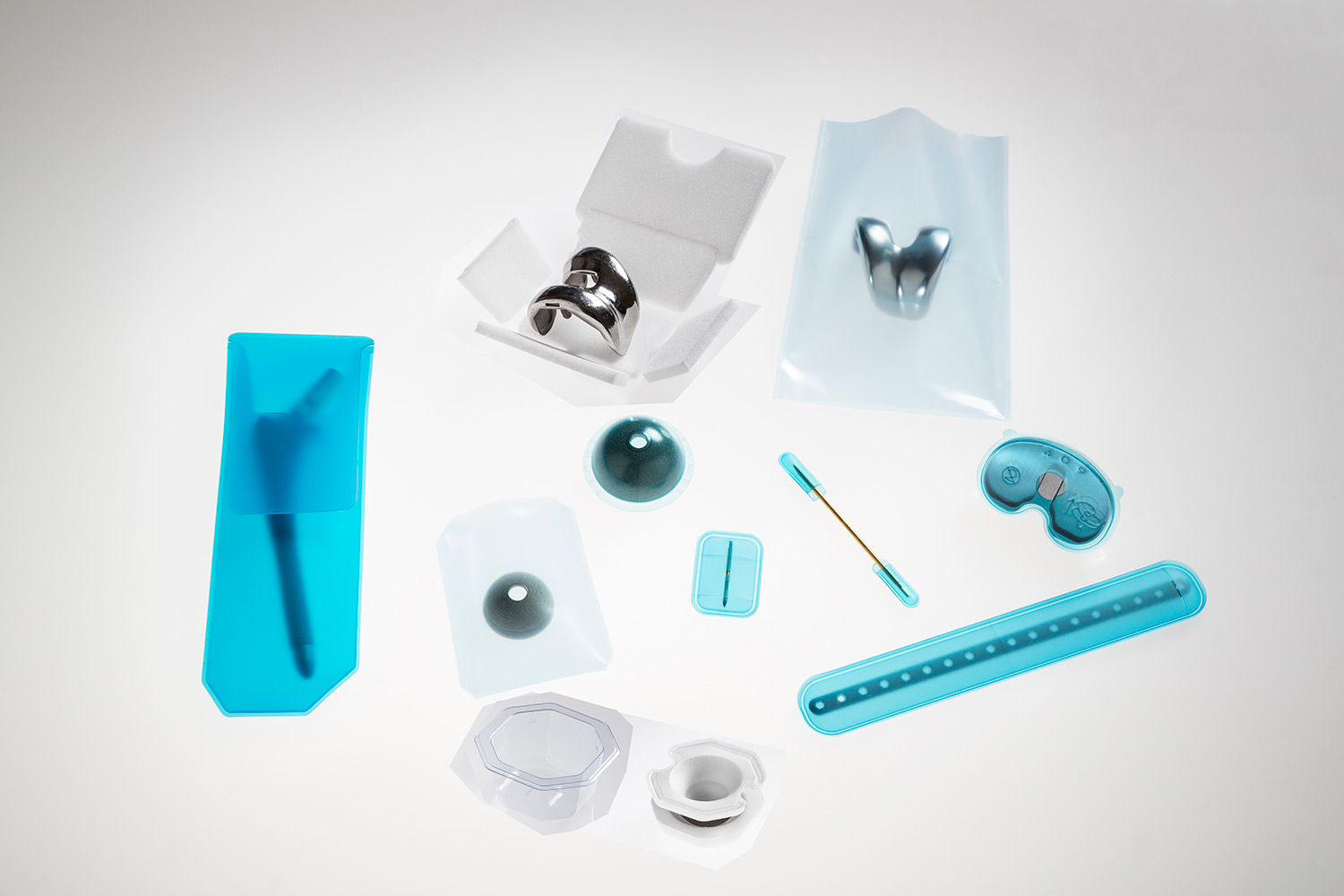
Applications
- Fusion devices
- Pedicle screws
- Plates
- Spacers
- Fixations
- Tools
Capabilities
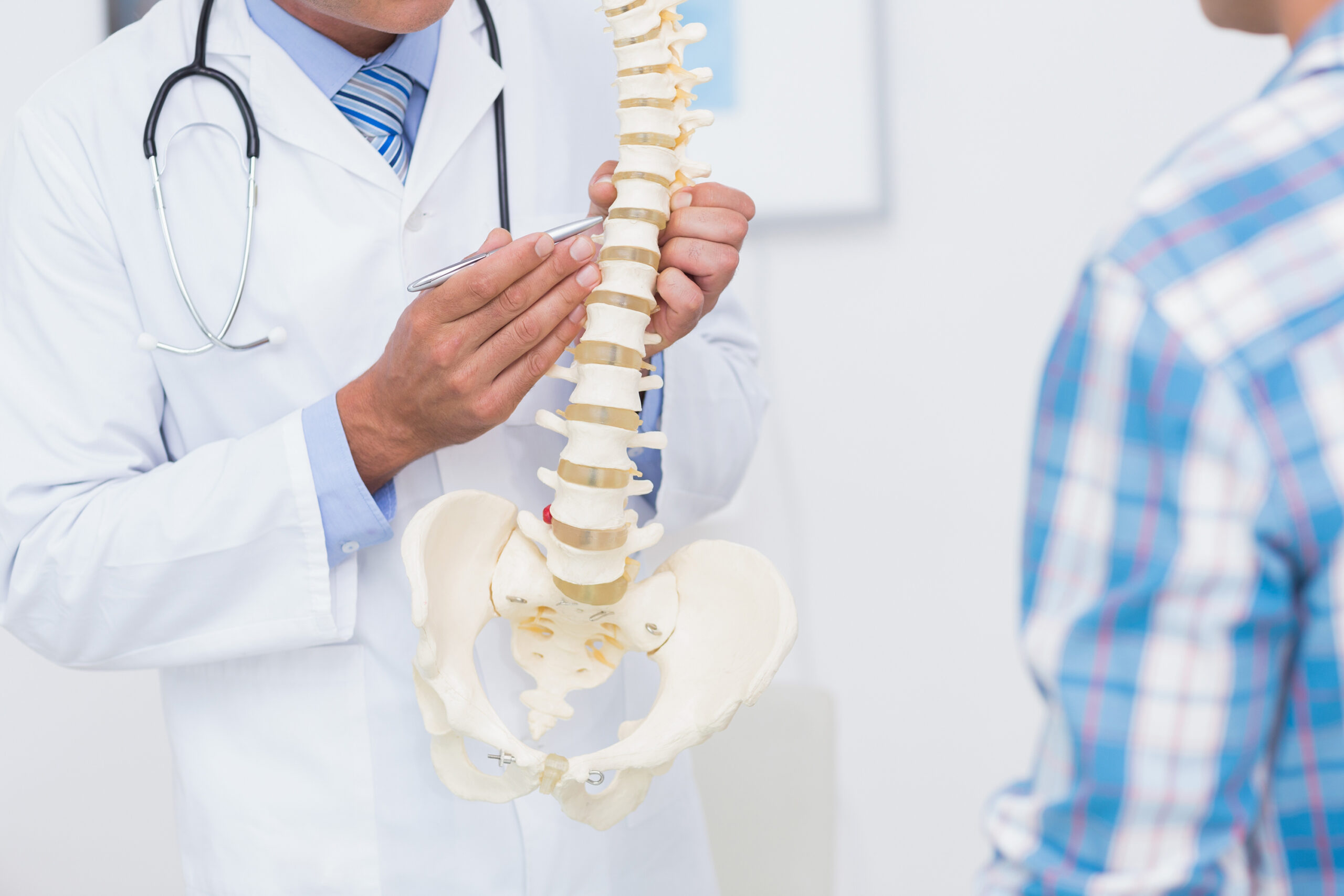
When a sterile spinal implant is ready for surgery, there can be no question about the integrity of its packaging. At UFP MedTech, we specialize in providing both standard and custom sterile barrier packaging for spinal implants including fusion devices, pedicle screws, plates, spacers, fixations, and tools. Our industry proven, USP Class VI, FDA approved FlexShield® TPU packaging and chemically pure medical grade foams are ideal for aseptic presentation in the operating room.
For decades, UFP MedTech has created the industry’s most innovative packaging solutions using the world’s most advanced materials to provide sterile barrier integrity protection throughout the supply chain.
Our spinal packaging systems can reduce costs and inventory by providing you the flexibility to consolidate part numbers by incorporating multiple products within a single protective package.
Materials
UFP MedTech has access to a range of advanced medical grade materials. These materials are compatible with multiple sterilization methods and offer excellent puncture and abrasion resistance. The following materials are commonly used for creating our spinal implant packaging solutions:
FlexShield® Thermoplastic Polyurethane Films (TPU)
FlexShield is made from a flexible thermoplastic polyurethane film. Its durability and sterility have made it the material of choice for orthopedic implants as well as protection for surgical instruments and equipment.
- USP Class VI resin and FDA approval
- Abrasion and puncture resistant
- Antimicrobial and biocompatible
- Gamma and EtO sterilization compatible
- Chemically resistant and inert
Medical Foams
In use for decades in orthopedic packaging, our cross-linked polyethylene (XLPE) packaging is a chemically inert, non-toxic, pure white foam.
- Lightweight and durable
- Impact resistant
- Gamma and Eto sterilization compatible
- Low odor and no outgassing

Request a Consultation